 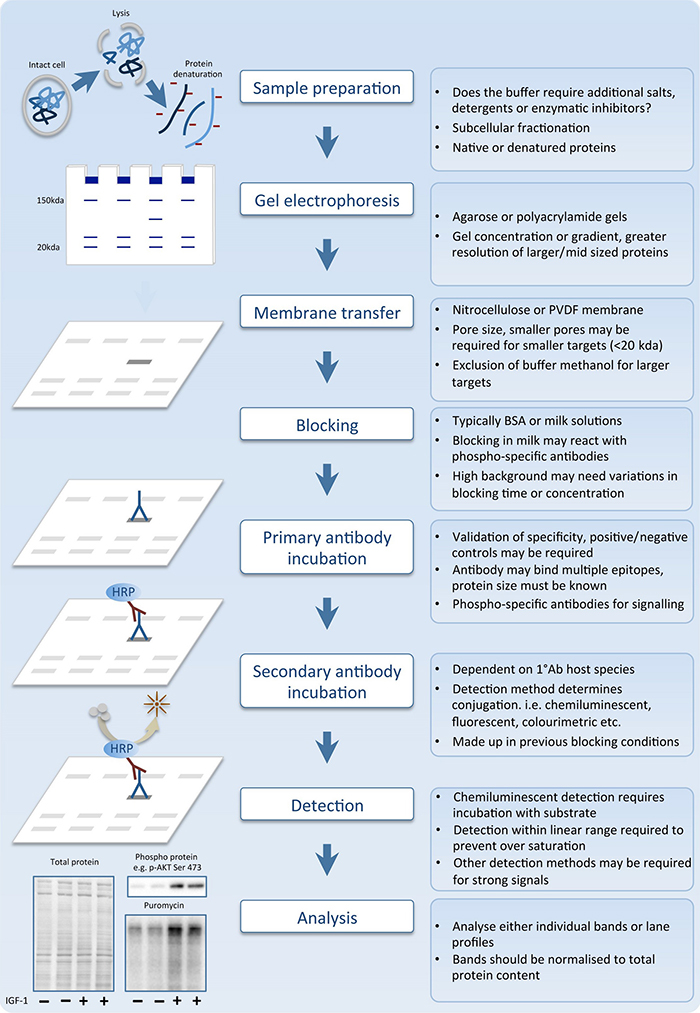
March 1990, FDA decided not to recommend routine anti-HIV-2īlood donated for transfusion. Public discussion at the FDA Blood Products Advisory Committee Raised the possibility of routine donor screening for HIV-2. Purification of HIV-2 grown in cell culture. Genetic Systems HIV-2 EIA, manufactured and distributed by GeneticĬorp., Redmond, WA, is based on a disrupted whole-virus antigen On April 25, 1990, the Food and DrugĪdministration (FDA) licensed an enzyme immunoassay (EIA) test kitĭetection of antibodies to HIV-2 in human serum or plasma. Because HIV-2Īre not always detected by HIV-1 antibody tests (4), antibody tests Relatively common in parts of West Africa (1-3). Immunodeficiency virus type 2 (HIV-2), was discovered and found to Second virus causing the acquired immunodeficiency syndrome (AIDS), (HIV-1), particularly through the blood supply, led to the rapidĭevelopment in 1985 of diagnostic tests for HIV-1 antibodies. If the HIV-2 EIA is positive, an HIV-2 supplemental testĮfforts to prevent transmission of human immunodeficiency Western blot result is negative or indeterminate, an HIV-2 EIA HIV-2 is recommended only if HIV-2 risk factors are present. Western blot confirms the presence of antibodies to HIV, and 
(or another licensed HIV-2 supplemental test). Reactive specimen by HIV-1/HIV-2 EIA should be tested by HIV-1 HIV-2 is performed by means of a combination HIV-1/HIV-2 EIA. 
The following procedures are recommended if testing for both Infection are present, if clinical evidence exists for HIV diseaseĪbsence of a positive test for antibodies to HIV-1, or if HIV-1īlot results exhibit the unusual indeterminate pattern of gag plus However, when HIV testing is indicated, tests for antibodies toĪnd HIV-2 should be obtained if epidemiologic risk factors for Recommend routine testing for HIV-2 in settings other than blood Persons in the United States is extremely low. Guidelines for serologic testing with combination HIV-1/HIV-2Įpidemiologic data indicate that the prevalence of HIV-2 Persons being tested in settings other than blood centers and This articleĬDC recommendations for the diagnosis of HIV-1 and HIV-2 infections Type 2 (HIV-2) beginning no later than June 1, 1992. The Food and Drug Administration (FDA) has recommended thatĭonated blood be screened for antibodies to human immunodeficiency This report was prepared by the following: Please note: This guideline document is obsolete and may not reflect current evidence or best practice and likely contains out-of-date information. Testing for Antibodies to Human Immunodeficiency Virus Type 2 For assistance, please send e-mail to: Type 508 Accommodation and the title of the report in the subject line of e-mail. Persons using assistive technology might not be able to fully access information in this file. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
